Key Takeaways
|
Understanding Manufacturing Audits
- Internal audits (first-party): Self-assessments run by the company to verify SOP compliance and readiness.
- Supplier or customer audits (second-party): Reviews by customers to verify capability, consistency, and controls.
- External audits (third-party): Independent certification or regulatory audits such as ISO certification, FDA-style inspections, or OSHA-style safety audits.
The Strategic Value of Audit Readiness
Audit readiness creates business value by reducing operational risk and strengthening trust with customers, lenders, and regulators. A strong audit history acts like reputation capital because it signals that controls are working, not just documented.
Teams often discover hidden inefficiencies during audit prep because they must prove that processes are consistent, measurable, and repeatable. When those gaps are corrected, plants typically see fewer disruptions, less rework, and cleaner handoffs across departments.
Audit preparation often requires SOP reviews and evidence checks that reveal duplicated steps, outdated approvals, and unclear responsibilities. Fixing those issues reduces process friction and lowers the risk of costly errors that result in scrap, rework, or shipment holds.
- Review and align SOPs with actual work instructions on the floor.
- Remove obsolete versions from active use and enforce document control.
- Confirm that preventive maintenance and calibration evidence is complete and traceable.
Many global supply chains require certifications and clean audit outcomes to qualify suppliers. Passing audits smoothly helps sales conversations by demonstrating reliability, disciplined execution, and consistent product quality.
Comprehensive Audit Preparation Checklist
Preparation is the antidote to panic. The following checklist is designed to be exhaustive, covering the critical areas that auditors invariably scrutinize. While specific requirements will vary based on the industry (e.g., aerospace vs. food processing), these core pillars remain constant across the manufacturing spectrum.
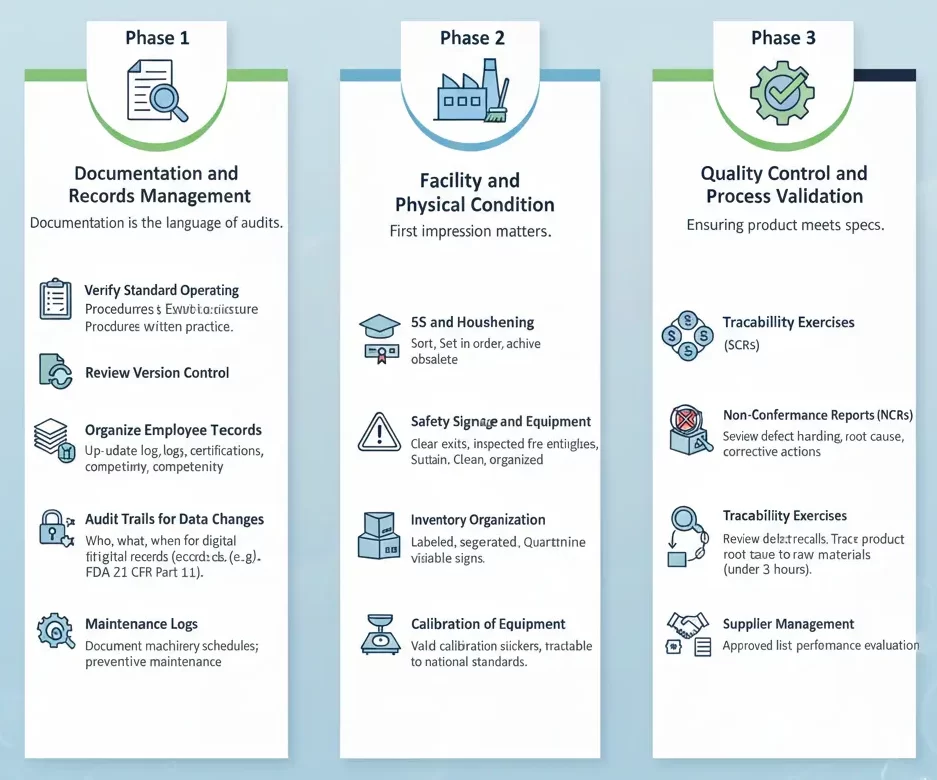
Phase 1: Documentation and Records Management
Documentation is the language of audits; if it is not written down, in the eyes of an auditor, it did not happen. This phase usually consumes the most time and requires the highest attention to detail.
- Verify Standard Operating Procedures (SOPs): Ensure that every critical process on the production floor has a corresponding written SOP. More importantly, verify that the current practice matches the written procedure. Discrepancies between “what is written” and “what is done” are the most common source of non-compliance findings.
- Review Version Control: Check that all documents are the current versions. Obsolete documents should be archived and removed from the active workspace to prevent confusion.
- Organize Employee Training Records: Auditors will verify if employees are qualified for their roles. Ensure that training logs, certification renewals, and competency assessments are up-to-date and easily accessible.
- Audit Trails for Data Changes: For digital records, ensure there is a clear history of who accessed data, what changes were made, and when. This is particularly critical for industries regulated by standards like FDA 21 CFR Part 11.
- Maintenance Logs: Ensure that all machinery maintenance schedules are documented. Proof of preventive maintenance is essential for demonstrating equipment reliability and safety.
Phase 2: Facility and Physical Condition
An auditor’s first impression is formed the moment they walk onto the factory floor. A disorganized facility suggests a disorganized management system.
- 5S and Housekeeping: Implement or refresh 5S (Sort, Set in order, Shine, Standardize, Sustain) methodologies. Aisles should be clear, tools should be organized, and the facility should be clean.
- Safety Signage and Equipment: Verify that all emergency exits are unobstructed, fire extinguishers are inspected, and safety signage (e.g., “Hard Hat Area”) is visible and legible.
- Inventory Organization: Raw materials, work-in-progress, and finished goods should be clearly labeled and segregated. Quarantine areas for non-conforming products must be strictly controlled to prevent accidental use.
- Calibration of Equipment: Check that all measuring and monitoring devices (scales, thermometers, calipers) have valid calibration stickers and that the master calibration records are traceable to national standards.
Phase 3: Quality Control and Process Validation
This section addresses the core of manufacturing: ensuring the product meets the required specifications.
- Non-Conformance Reports (NCRs): Review the history of NCRs. Auditors will look at how you handled defects. Did you identify the root cause? Did you implement corrective actions? Is there evidence that the corrective action was effective?
- Traceability Exercises: Conduct mock recalls. Pick a finished product at random and see if you can trace it back to the specific raw material batches used, including the suppliers and intake dates, within a short timeframe (typically under 4 hours).
- Supplier Management: Ensure that your approved supplier list is current and that you have evidence of evaluating your suppliers’ performance.
The Role of Technology in Audit Readiness
The era of preparing for audits with binders full of paper and dusty filing cabinets is rapidly fading. The Role of Technology has shifted from being a supportive element to a central pillar of audit readiness. Modern manufacturing environments utilize integrated software solutions to maintain a state of “continuous auditability.”
This approach uses Enterprise Resource Planning (ERP) systems to centralize data, ensuring a single source of truth for all operational metrics.
Automation of Compliance Data
Manual data entry is prone to human error a transposed number or a missing signature can lead to a compliance finding. Advanced manufacturing software automates the collection of data directly from machines and IoT sensors.
For instance, instead of an operator manually logging the temperature of a pasteurization tank every hour, smart sensors can log the data continuously to a secure cloud database. This not only ensures 100% data accuracy but also provides auditors with granular, irrefutable evidence of process control.
Centralized Document Management
One of the biggest challenges during an audit is retrieving specific documents requested by the auditor. Searching through physical archives is time-consuming and stressful. Digital solutions allow manufacturers to tag, index, and store documents in a centralized repository.
With a few keystrokes, a manager can pull up maintenance records for a specific machine from three years ago or training certificates for a specific shift team. Solutions provided by various technology vendors, including platforms like SAP, Oracle, and HashMicro, facilitate this level of digital organization, drastically reducing the administrative burden during inspections.
Common Pitfalls to Avoid
Facilities usually fail audits due to predictable behavior patterns, not because requirements are unclear. Avoiding these pitfalls improves audit outcomes and also improves day-to-day control quality.
- The audit prep panic: Last-minute fixes encourage risky shortcuts, such as backfilling forms and rushing corrective actions without proper validation. Experienced auditors spot patterns, such as identical handwriting, across months of logs, which can escalate issues into integrity concerns.
- Siloed information: Audit findings often appear when maintenance, QA, and production act independently and controls do not connect. Cross-functional routines and shared systems help ensure that changes such as repairs, recalibrations, and release decisions are tracked and communicated.
- Neglecting the human element: Auditors interview operators, so knowledge gaps quickly become findings even when paperwork looks complete. Coaching should focus on providing truthful, precise answers and ensuring clear awareness of where the latest SOPs and safety rules are located.
Future Trends in Manufacturing Audits
As we look toward 2025 and beyond, the landscape of manufacturing audits is evolving. The integration of Industry 4.0 technologies is changing not just how manufacturers operate, but how they are audited.
Strategic Implementation: A Data-Driven Approach
An ERP implementation works best when it is treated as an operational transformation supported by software and measured outcomes. A phased rollout reduces disruption and helps teams correct issues before they spread across the business.
Phase 1 Process Mapping and Gap Analysis
Teams should document current workflows end-to-end and identify where controls break, where approvals are unclear, and where data becomes unreliable. Gap analysis then clarifies which processes can follow standard ERP best practices and which need justified adjustments.
Phase 2 Data Sanitation and Migration
Migration succeeds when data is governed early, because dirty data can damage trust in the system immediately after go-live. Clear governance rules prevent duplicates, inconsistent naming, and obsolete records from contaminating daily operations.
- De-duplication: Merge duplicate customer, supplier, and item records.
- Standardization: Align units of measure, naming conventions, and classification rules.
- Archiving: Separate historical compliance data from active transactional data.
Phase 3 Testing and User Acceptance Testing
Testing should simulate real workflows and edge cases so the system behaves correctly under operational pressure. UAT sign-off should happen only after role-based users can complete daily tasks confidently without workarounds.
Defining Success with Metrics and KPIs
To validate the ROI of an Equip ERP implementation, organizations must track specific Key Performance Indicators (KPIs) before and after go-live. These metrics provide objective evidence of operational improvement.
Operational Efficiency Metrics
- Order Cycle Time: Measure the time elapsed from order placement to delivery. A successfully implemented ERP should reduce this by automating credit checks, inventory allocation, and pick-ticket generation.
- Inventory Turnover Ratio: This metric indicates how often inventory is sold and replaced over a period. Equip ERP’s demand forecasting tools should lead to a higher turnover ratio, indicating efficient capital usage and reduced holding costs.
Financial Accuracy Metrics
- Days Sales Outstanding (DSO): By automating invoicing and dunning letters, the ERP should accelerate cash flow, reducing the average time it takes to collect payment.
- Cash-to-Cash Cycle Time: This comprehensive metric measures the time between paying for raw materials and getting paid for the finished product. Tight integration between Accounts Payable, Inventory, and Accounts Receivable modules in Equip ERP should compress this cycle significantly.
Quality and Compliance Metrics
- Perfect Order Rate: The percentage of orders that are on time, complete, undamaged, and with correct documentation. This is the ultimate measure of supply chain reliability.
- Schedule Compliance: For manufacturers, this measures how often the production floor adheres to the planned schedule. Deviations highlight issues in machine maintenance or raw material availability.
Common Pitfalls and Mitigation Strategies
Even with a robust system like Equip ERP, projects can falter due to human and organizational factors. Recognizing these pitfalls early is essential for course correction.
Scope Creep and Customization Overload
- The Pitfall: Stakeholders request excessive ERP customizations to replicate legacy systems, resulting in “paving the cow path” and automating inefficient processes. Over-customization also complicates upgrades and long-term system maintenance.
- The Mitigation: Apply a “Vanilla First” policy by requiring a clear business case for every customization request. Prioritize standard functionality in Equip ERP and reserve customization only for features that create real competitive advantage.
Inadequate Change Management
- The Pitfall: Projects focus too heavily on technical deployment while ignoring employee adoption, leading to resistance, low usage, and shadow IT.
- The Mitigation: Appoint Change Champions in each department to drive engagement and peer support. Deliver continuous, role-based training programs tailored to operational needs, not just one-time sessions.
Underestimating Data Complexity
- The Pitfall: Treating data migration as a last-minute task often causes delays, inaccuracies, and data corruption at go-live.
- The Mitigation: Manage data migration as a dedicated sub-project with its own timeline and resources. Start early data cleansing and use automated validation tools to ensure data integrity during transfer.
Advanced Best Practices for Long-Term Value
After stabilization, optimization becomes the priority because it turns the ERP into a control engine rather than a record-keeping tool. Advanced practices work best when they focus on exceptions and prevention.
- Automated Workflow Triggers
Exception-based triggers help managers focus on high-impact decisions rather than routine monitoring. Rules like margin thresholds, approval holds, and reorder alerts improve control discipline while reducing manual oversight. - Integration with IoT and Edge Computing
IoT integration improves timeliness by updating records on machine performance and warehouse movements without delay. This reduces reconciliation work and supports preventive maintenance before breakdowns occur. - Predictive Analytics and Demand Planning
Forecasting models improve purchasing and production planning when they incorporate seasonality, lead-time variability, and sales velocity. Strong demand planning reduces stockouts and excess inventory while supporting more reliable schedules.
After these best practices are in place, ERP Manufacturing can help manufacturers sustain them at scale by keeping workflows, approvals, and operational records connected in one system. With centralized data, clearer audit trails, and configurable controls, teams can manage exceptions faster, reduce manual follow-ups, and maintain consistent execution across shifts and sites, so optimization becomes a stable routine rather than a one-off initiative.
Conclusion
Audit preparation is a continuous discipline that reinforces operational control, data accuracy, and workplace safety. When readiness becomes part of daily routines rather than a last-minute effort, audits are more predictable and far less disruptive.
A structured framework organizes preparation into clear phases, defined evidence standards, and accountable ownership across teams. Supported by the right technology, this approach improves traceability, accelerates document retrieval, and strengthens the reliability of audit trails.
As audit expectations continue to evolve, manufacturers gain an advantage by investing in digital documentation and measurable internal controls early on. To understand how an integrated ERP system can support audit-ready workflows and stronger traceability, consider consulting with a specialist to evaluate your current processes and gaps.
FAQ About Manufacturing Audit Preparation
-
How often should a manufacturing facility conduct internal audits?
Internal audits are typically conducted once or twice a year, and more frequently for high-risk processes or areas with recurring findings. Regular cadence helps the facility stay continuously ready and reduces the chance of last-minute gaps before external inspections.
-
What is the difference between a first-party and a third-party audit?
A first-party audit is an internal assessment performed by the company’s own team to verify process compliance and identify improvement opportunities. A third-party audit is conducted by an independent body or regulator to confirm compliance with a standard or requirement such as ISO certification or regulatory expectations.
-
How can technology improve audit preparation?
Technology reduces manual effort by centralizing documents, enforcing version control, and creating reliable audit trails for approvals and data changes. It also speeds up evidence retrieval during the audit, which lowers stress and improves consistency across teams.
-
What are the most common reasons for failing a manufacturing audit?
Common causes include missing or incomplete documentation, gaps between written SOPs and actual practices, outdated document versions in use, and weak training records. Poor housekeeping, unclear maintenance logs, and inconsistent corrective action evidence also frequently lead to findings.
-
What is a mock recall and why is it important for audits?
A mock recall is a traceability test where the facility selects a finished product and proves it can trace back to raw material lots, suppliers, and processing records within a defined timeframe. It validates that traceability controls are working, which is critical for quality and safety audits in regulated industries.